 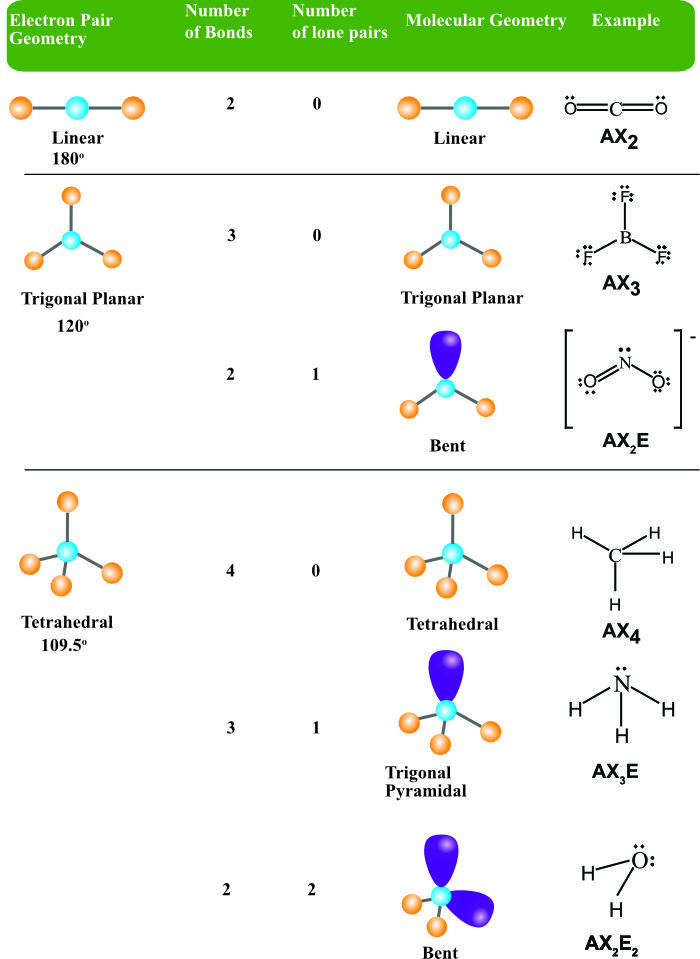
More.Ĭv::findCirclesGrid ( InputArray image, Size patternSize, OutputArray centers, int flags= CALIB_CB_SYMMETRIC_GRID, const Ptr &blobDetector= SimpleBlobDetector::create())Ĭv::findEssentialMat ( InputArray points1, InputArray points2, InputArray cameraMatrix, int method= RANSAC, double prob=0.999, double threshold=1.0, int maxIters=1000, OutputArray mask= noArray())Ĭalculates an essential matrix from the corresponding points in two images. Hirschmuller algorithm that differs from the original one as follows: More.Ĭv::CALIB_USE_LU = (1 &blobDetector, const CirclesGridFinderParameters ¶meters)įinds centers in the grid of circles. The base class for stereo correspondence algorithms. But as the structure of Ozone has resonance and one lone pair of electrons, the angle between the molecules is less than 120 degrees.Class for computing stereo correspondence using the block matching algorithm, introduced and contributed to OpenCV by K. Ozone has sp2 hybridization means that it should have a trigonal planar shape. O3 Molecular GeometryĪs the hybridization of the molecule determines its shape, we can now know the molecular geometry of Ozone. As we always consider the hybridization of the central atom as the final hybridization, Ozone has sp2 hybridization. One will have sp2 hybridization, whereas, the other will have sp3 hybridization as there is one lone pair of electrons that creates resonance in the structure of Ozone. Other two oxygen atoms also have hybridization. 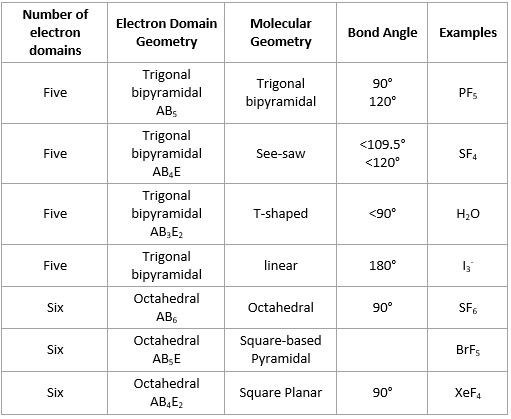
As there are electrons in one s orbital, and two p orbitals, the hybridization of the central oxygen atom becomes sp2. There are two electrons in the 2s orbital, whereas 6 electrons in both the 2p orbitals out of three 2p orbitals. As Ozone has one central Oxygen atom having eight electrons in its outermost shell, the hybridization for the central atom will be sp2. 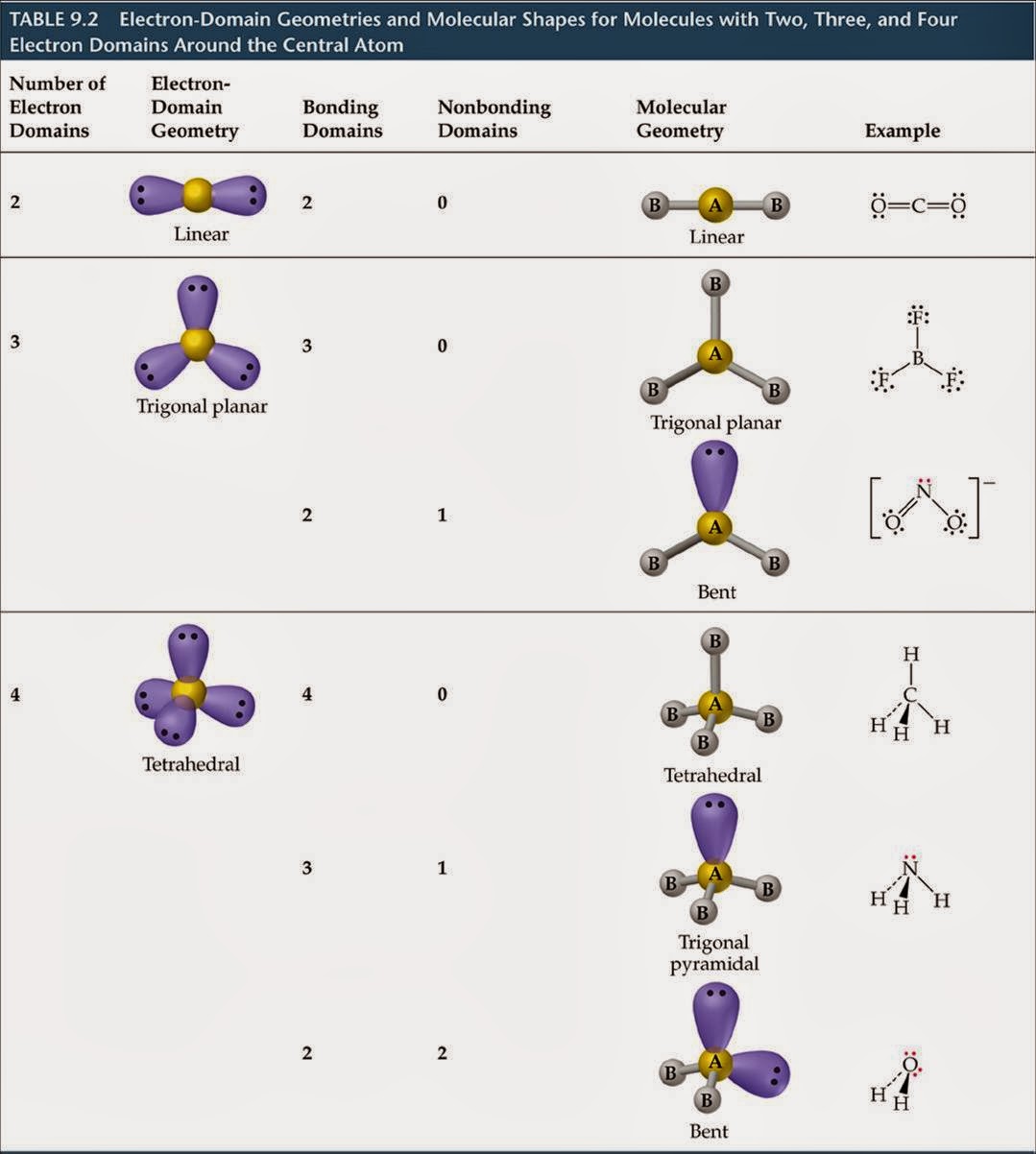
Once we know the Lewis structure of a molecule, it is easy to know the hybridization of it. Hybridization in chemistry means the hybridising of two or more atomic levels of the same or different energies to combine and give a new orbital. As both the molecules of Oxygen have the same electronegativity and structure, the double bond keeps on shifting from both the molecules. To satisfy the octet rule, a central atom needs to form a double bond on either of its sides with an Oxygen molecule and another single bond. 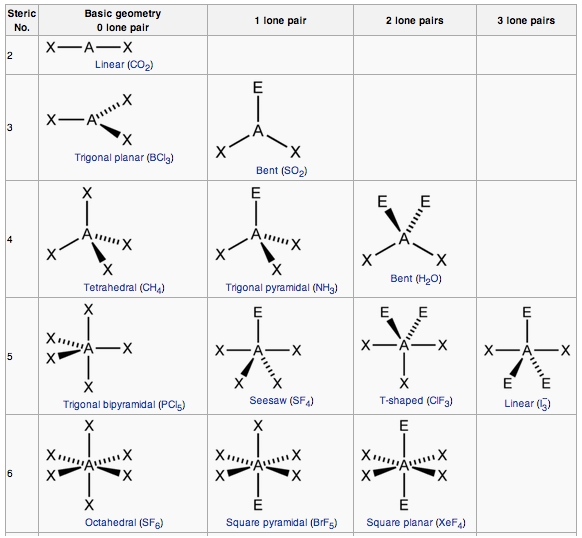
The central atom has one lone pair of electrons and is stable due to the eight electrons in its outermost orbit. So one molecule of the Oxygen is in the centre with the other two are on the opposite sides. O3 Lewis StructureĪs the octet rule applies in this structure, the central atom is the first one that should have eight electrons in its outer shell. Thus there are a total of 18 valence electrons available for Ozone molecule. Here as there are three oxygen molecules, the total number of valence electrons is 6*3= 18. In Ozone or O3, there are six valence electrons for each molecule of Oxygen. O3 Polar or Nonpolar O3 Valence electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
